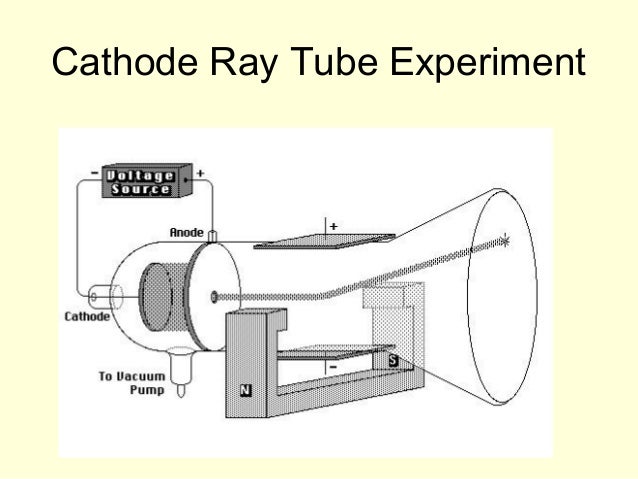
Thomson discovered the first subatomic particle, the electron, while researching cathode rays. Johnstone Stoney to denote the unit of charge found in experiments that passed electrical current through chemicals it was Irish physicist George Francis Fitzgerald who suggested in 1897 that the term be applied to Thomson’s corpuscles.) What is the atomic model of JJ Thomson? (The term “electron” was coined in 1891 by G. His work also led to the invention of the mass spectrograph. In 1897 Thomson discovered the electron and then went on to propose a model for the structure of the atom. He placed cathode tubes in electric and magnetic fields. Thomson was a physics professor at Cambridge University in the UK. His research in cathode rays led to the discovery of the electron, and he pursued further innovations in atomic structure exploration. Thomson attended Trinity College at Cambridge, where he would come to head the Cavendish Laboratory. Where did JJ Thomson discovered the electron? He named his discovery “protons” based on the Greek word “protos” which means first. During this period, his research resulted in a nuclear reaction which led to the first ‘splitting’ of the atom, where he discovered protons. The proton was discovered by Ernest Rutherford in the early 1900’s. 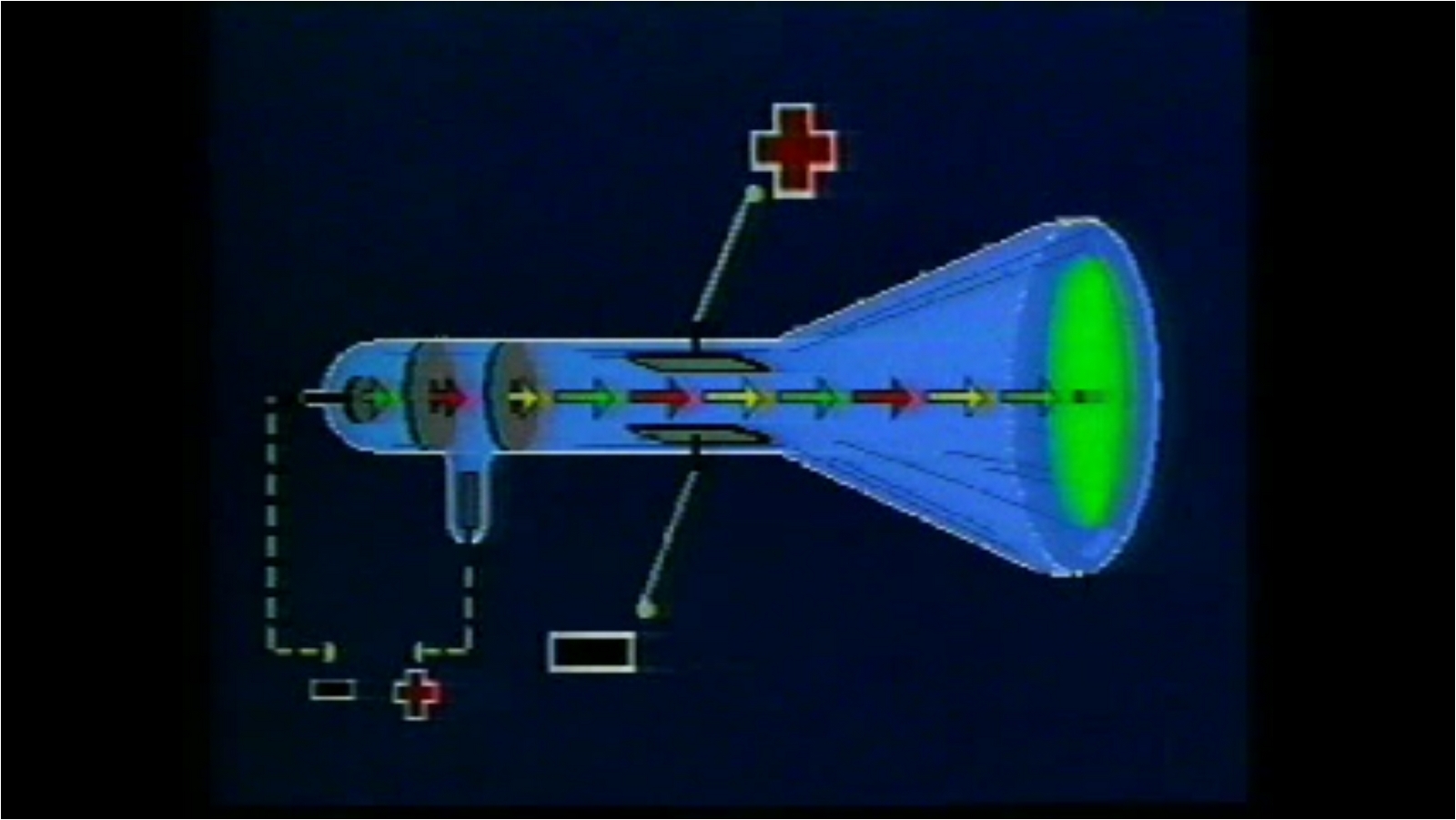
One of Thomson’s students was Ernest Rutherford, who later succeeded him as Cavendish Professor of Physics. During his experiment he discovered electrons and it is one of the most important discoveries in the history of physics. Thomson experimenting with cathode ray tubes. The Cathode ray experiment was a result of English physicists named J. Thomson concluded that neon exists in two forms whose masses are different – i.e. he used a mass spectrometer – and observed two distinct deflections. Thomson made this discovery when his research student Francis Aston fired ionized neon through a magnetic and electric field – i.e. They were too small to have their mass or charge calculated directly, but he attempted to deduce this from how much the particles were bent by electrical currents, of varying strengths. He decided to try to work out the nature of the particles. Thomson, in full Sir Joseph John Thomson, (born December 18, 1856, Cheetham Hill, near Manchester, England-died August 30, 1940, Cambridge, Cambridgeshire), English physicist who helped revolutionize the knowledge of atomic structure by his discovery of the electron (1897). Thomson discovered negatively charged particles by cathode ray tube experiment in the year 1897. Thomson was the first and one of the many scientists who proposed models for the structure of an atom. In addition, he also studied positively charged particles in neon gas. He demonstrated that cathode rays were negatively charged. Thomson discovered the electron by experimenting with a Crookes, or cathode ray, tube.  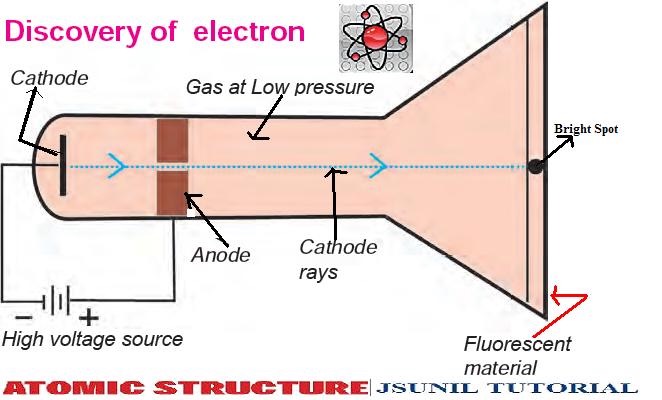
Poynting in a four-volume textbook of physics, Properties of Matter and in 1895 he produced Elements of the Mathematical Theory of Electricity and Magnetism, the 5th edition of which appeared in 1921. Thomson atomic model, earliest theoretical description of the inner structure of atoms, proposed about 1900 by William Thomson (Lord Kelvin) and strongly supported by Sir Joseph John Thomson, who had discovered (1897) the electron, a negatively charged part of every atom. This finding revolutionized the way scientists thought about the atom and had major ramifications for the field of physics. Thomson announced his discovery that atoms were made up of smaller components. On April 30, 1897, British physicist J.J. 24 Who split the first atom? What did Thomson discover?
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
